Properties of water
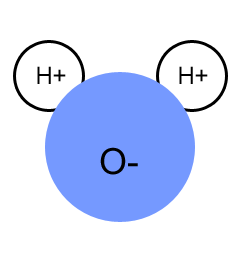
Water is two parts of hydrogen & part oxygen
An .Atom is the smallest Unit of matter that retains retains its properties. A
Atoms consist of Protons, neutrons, & Electrons.
Protons have a positive change. Electrons Have a negative charge, & Neutrons have no charge no charge (Neutral) .
Atoms are usually neutrally charged because of the number of protons Are even. Unevenly charged atoms are called ions.
Water Is a polar molecule. Shared electrons spend more time on the O than the H.
Hydrogen Bonding - Weak attraction between weakly positive hydrogen atoms & weakly negative oxygen atoms of different molecule.
States Of Matter
solid,liquid,&Gas.
Temperature- Substance is a measure of average kinetic energy of the atoms & Molecules.
Heat - measure or the total kinetic energy of the atoms of molecules(measured in calories)
One calorie is equal to the amount of heat needed to raise the temperature.
Density- Is A Physical property of a substance that describes the degree of compactness of a substance in other words How closely packed together the atoms are. (The more closely the atoms are in a substance the more dense it is).
An .Atom is the smallest Unit of matter that retains retains its properties. A
Atoms consist of Protons, neutrons, & Electrons.
Protons have a positive change. Electrons Have a negative charge, & Neutrons have no charge no charge (Neutral) .
Atoms are usually neutrally charged because of the number of protons Are even. Unevenly charged atoms are called ions.
Water Is a polar molecule. Shared electrons spend more time on the O than the H.
Hydrogen Bonding - Weak attraction between weakly positive hydrogen atoms & weakly negative oxygen atoms of different molecule.
States Of Matter
solid,liquid,&Gas.
Temperature- Substance is a measure of average kinetic energy of the atoms & Molecules.
Heat - measure or the total kinetic energy of the atoms of molecules(measured in calories)
One calorie is equal to the amount of heat needed to raise the temperature.
Density- Is A Physical property of a substance that describes the degree of compactness of a substance in other words How closely packed together the atoms are. (The more closely the atoms are in a substance the more dense it is).