How does carbon exist in the atmosphere?
Carbon exists in a chemical compound with oxygen: Carbon dioxide (CO2)
How are fossil fuels created?
The carbon found in decaying plants and animals is trapped underground for millions of years
and overtime becomes coal (or other fossil fuels).
Describe two ways that carbon enters the atmosphere.
Carbon can enter the atmosphere when cells convert sugar into energy (that process is called
cellular respiration). When we burn fossil fuels, we’re releasing stored carbon into the air.
How are the oceans involved in the carbon cycle?Carbon dioxide from the atmosphere dissolves in the surface waters of the ocean. Some of the carbon dioxide stays as dissolved gas, but much of it gets turned into other things. Photosynthesis by tiny marine plants (phytoplankton) in the sunlit surface waters turns the carbon into organic matter
What role do rocks have within the carbon cycle?
Over time, carbon can create rocks like limestone, dolomite, calcite, and marble. This carbon is
then “trapped” within the rock for a long time. It can gradually be removed from the rock as
water reacts with it (in, for example, the formation of a cave).
Where are you starting within the carbon cycle?
You’re starting with the atmosphere.
. How much of the atmosphere is made of carbon dioxide (CO2)?
Carbon dioxide makes up 0.04% of the atmosphere.
By how much has CO2 increased in the atmosphere during the past 150 years?
It has increased by 30% during the past 150 years.
Next stop = ____Surface Ocean_____________________________________
What did you learn:The deep ocean accounts for more
than _65__ % of
the Earth’s
carbon
Next stop = ___Yellow Star______________________________________
What did you learn? Only 0.04% of the atmosphere is carbon dioxide. A little can make a huge impact on global warming!
Next stop = _Yellow Star________________________________________
What did you learn?5,000 Megatons of CO2 is produced each year by burning fossil fuels. That's a lot of carbon!
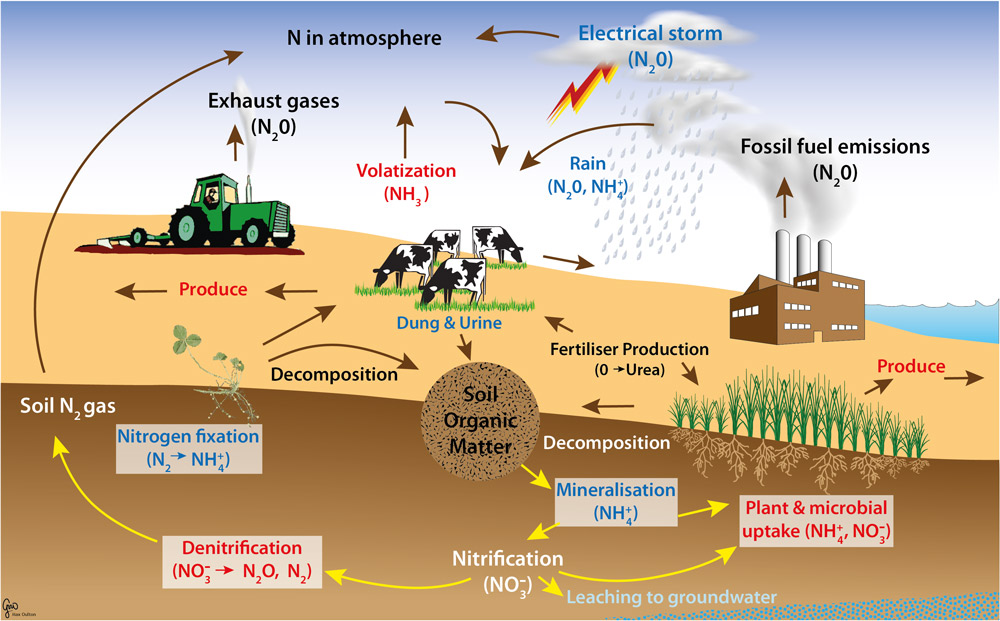
What are the two conditions under which nitrogen will react with oxygen? (In other words,
what is necessary for nitrogen in the air to combine with oxygen?)Nitrogen will react with oxygen in conditions with really high temperatures and pressures.
This might happen during lightning.
What are the two compounds that are formed when nitrogen combines with oxygen?
NO and NO2
How does nitric acid (HNO3) form?
Nitric acid forms when NO2 reacts with water.
Why is nitric acid (HNO3) important?
Nitric acid can be used by plants to obtain the nitrogen they need.
What percentage of the air we breathe is nitrogen?
The air we breathe is 79% nitrogen.
Even though considerable nitrogen is available in the air, most plants do not use the nitrogen
(N2) found in the air. Why not?
Plants are not able to use nitrogen in this form.
In what compounds can plants use nitrogen?
Plants can only use nitrogen when it has been converted or “fixed” into one of these
compounds: NO3, NH3, or (NH2)2CO
How do animals get the nitrogen they need?
Animals can only get nitrogen by eating plants or eating other animals (who got their nitrogen
by eating plants).
What percentage of the air we breathe is nitrogen?
The air we breathe is 79% nitrogen.
Even though considerable nitrogen is available in the air, most plants do not use the nitrogen
(N2) found in the air. Why not?
Plants are not able to use nitrogen in this form.
In what compounds can plants use nitrogen?
Plants can only use nitrogen when it has been converted or “fixed” into one of these
compounds: NO3, NH3, or (NH2)2CO
How do animals get the nitrogen they need?
Animals can only get nitrogen by eating plants or eating other animals (who got their nitrogen
by eating plants).
Atmospheric nitrogen (N2) is pretty inert. This means that it does not easily break apart. When
molecules do not break apart easily, it is difficult (or impossible) for organisms to use them as a
nutrient source. As a result, nitrogen fixation is the term used to describe the process of
breaking up N2.
a. What is atmospheric fixation?
In atmospheric fixation, the unusable nitrogen found in the atmosphere is converted into
a form useable by plants. That process occurs under incredibly high temperatures
(during lightning). The newly created compounds reach earth/soil by dissolving in
water.
This creates only a small percentage (5-8%) of all the nitrogen useable by plants.
b. What is industrial fixation? [This is how artificial fertilizers are made.]
In industrial fixation, nitrogen is converted to a useable form by a chemical reaction that
takes nitrogen from the atmosphere and hydrogen from a fossil fuel in high heat to form
nitrogen compounds that plants can use.
c. What is biological fixation? (In your answer, describe the types of plants associated
with the symbiotic relationship.)
In biological fixation, a living organism (nitrogen-fixing bacteria) are able to convert
atmospheric nitrogen into a nitrogen-containing compound that plants can use. These
nitrogen-fixing bacteria live in a mutualistic relationship with legumes (bean-type
plants).
Draw the nitrogen cycle:
Carbon exists in a chemical compound with oxygen: Carbon dioxide (CO2)
How are fossil fuels created?
The carbon found in decaying plants and animals is trapped underground for millions of years
and overtime becomes coal (or other fossil fuels).
Describe two ways that carbon enters the atmosphere.
Carbon can enter the atmosphere when cells convert sugar into energy (that process is called
cellular respiration). When we burn fossil fuels, we’re releasing stored carbon into the air.
How are the oceans involved in the carbon cycle?Carbon dioxide from the atmosphere dissolves in the surface waters of the ocean. Some of the carbon dioxide stays as dissolved gas, but much of it gets turned into other things. Photosynthesis by tiny marine plants (phytoplankton) in the sunlit surface waters turns the carbon into organic matter
What role do rocks have within the carbon cycle?
Over time, carbon can create rocks like limestone, dolomite, calcite, and marble. This carbon is
then “trapped” within the rock for a long time. It can gradually be removed from the rock as
water reacts with it (in, for example, the formation of a cave).
Where are you starting within the carbon cycle?
You’re starting with the atmosphere.
. How much of the atmosphere is made of carbon dioxide (CO2)?
Carbon dioxide makes up 0.04% of the atmosphere.
By how much has CO2 increased in the atmosphere during the past 150 years?
It has increased by 30% during the past 150 years.
Next stop = ____Surface Ocean_____________________________________
What did you learn:The deep ocean accounts for more
than _65__ % of
the Earth’s
carbon
Next stop = ___Yellow Star______________________________________
What did you learn? Only 0.04% of the atmosphere is carbon dioxide. A little can make a huge impact on global warming!
Next stop = _Yellow Star________________________________________
What did you learn?5,000 Megatons of CO2 is produced each year by burning fossil fuels. That's a lot of carbon!
What are the two conditions under which nitrogen will react with oxygen? (In other words,
what is necessary for nitrogen in the air to combine with oxygen?)Nitrogen will react with oxygen in conditions with really high temperatures and pressures.
This might happen during lightning.
What are the two compounds that are formed when nitrogen combines with oxygen?
NO and NO2
How does nitric acid (HNO3) form?
Nitric acid forms when NO2 reacts with water.
Why is nitric acid (HNO3) important?
Nitric acid can be used by plants to obtain the nitrogen they need.
What percentage of the air we breathe is nitrogen?
The air we breathe is 79% nitrogen.
Even though considerable nitrogen is available in the air, most plants do not use the nitrogen
(N2) found in the air. Why not?
Plants are not able to use nitrogen in this form.
In what compounds can plants use nitrogen?
Plants can only use nitrogen when it has been converted or “fixed” into one of these
compounds: NO3, NH3, or (NH2)2CO
How do animals get the nitrogen they need?
Animals can only get nitrogen by eating plants or eating other animals (who got their nitrogen
by eating plants).
What percentage of the air we breathe is nitrogen?
The air we breathe is 79% nitrogen.
Even though considerable nitrogen is available in the air, most plants do not use the nitrogen
(N2) found in the air. Why not?
Plants are not able to use nitrogen in this form.
In what compounds can plants use nitrogen?
Plants can only use nitrogen when it has been converted or “fixed” into one of these
compounds: NO3, NH3, or (NH2)2CO
How do animals get the nitrogen they need?
Animals can only get nitrogen by eating plants or eating other animals (who got their nitrogen
by eating plants).
Atmospheric nitrogen (N2) is pretty inert. This means that it does not easily break apart. When
molecules do not break apart easily, it is difficult (or impossible) for organisms to use them as a
nutrient source. As a result, nitrogen fixation is the term used to describe the process of
breaking up N2.
a. What is atmospheric fixation?
In atmospheric fixation, the unusable nitrogen found in the atmosphere is converted into
a form useable by plants. That process occurs under incredibly high temperatures
(during lightning). The newly created compounds reach earth/soil by dissolving in
water.
This creates only a small percentage (5-8%) of all the nitrogen useable by plants.
b. What is industrial fixation? [This is how artificial fertilizers are made.]
In industrial fixation, nitrogen is converted to a useable form by a chemical reaction that
takes nitrogen from the atmosphere and hydrogen from a fossil fuel in high heat to form
nitrogen compounds that plants can use.
c. What is biological fixation? (In your answer, describe the types of plants associated
with the symbiotic relationship.)
In biological fixation, a living organism (nitrogen-fixing bacteria) are able to convert
atmospheric nitrogen into a nitrogen-containing compound that plants can use. These
nitrogen-fixing bacteria live in a mutualistic relationship with legumes (bean-type
plants).
Draw the nitrogen cycle: